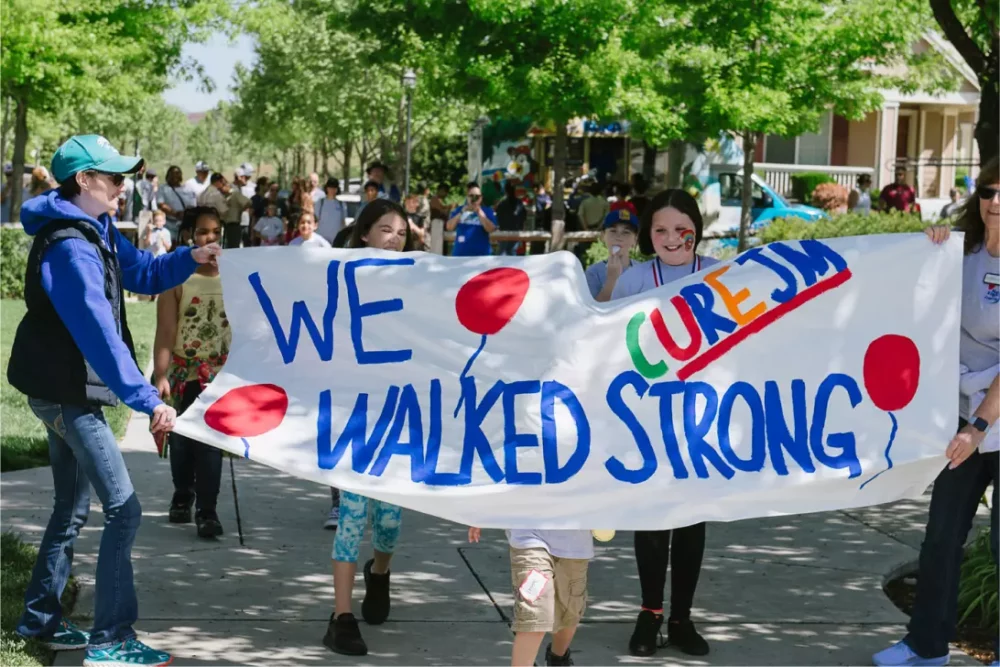Cure JM Events
Get involved with Cure JM throughout the year! Participating in events is a great way to move the mission forward and meet other JM families.

Town Halls
Cure JM Town Halls offer information on juvenile myositis treatments, research, social and emotional health for JM patients and families, and other pertinent topics related to JM.
Connects & Conversations
Cure JM invites you to join us for exciting events happening both online and in your community! Connect with other JM families at fun in-person regional gatherings, or take part in our many virtual events from the comfort of your home.
2026 Walk Strong to Cure JM
Cure JM families and friends are joining forces to Walk Strong together. Join the hundreds of Cure JM families who have taken steps toward better treatments and a cure.
To join a walk near you, click below! Or start a walk in your area!
Educational Family Events
You are invited to join us and meet other Cure JM families at an upcoming educational event, family day, or conference in your area.
2027 National Family Conference
We are excited to announce that the Cure JM National Family Conference will be back in 2027!
Join us for three days with world-class JM researchers, engaging sessions for all, and social time to connect with new friends. The 16th Annual Cure JM National Family Conference is taking place June 24th – June 27th, 2027, location TBD.
We’re inviting all families affected by juvenile myositis. That means you’re invited!